Vaccine Vial Monitor (VVM)
Definition
A Vaccine Vial Monitor (VVM) is a thermochromic chemical indicator label affixed to vaccine vials by the manufacturer. It undergoes a permanent, irreversible, and predictable color change in response to cumulative time and temperature exposure, serving as a proxy indicator for the thermal degradation of the specific vaccine.
Mechanism of Action
- The active material contains a substituted diacetylene monomer.
- Upon exposure to heat, the monomer polymerizes into a dark-colored polymer.
- The reaction rate is temperature-dependent and precisely calibrated to match the thermal stability profile (thermolability) of the specific vaccine.
Classification (WHO PQS Standards)
VVMs are classified based on the number of days required for the monitor to reach its discard point when continuously exposed to a constant reference temperature of 37°C.
- VVM 2 (High Heat Sensitivity): Reaches discard point in 2 days at 37°C. Exclusively used for Oral Polio Vaccine (OPV).
- VVM 7 (Moderate Heat Sensitivity): Reaches discard point in 7 days at 37°C. Used for vaccines like Measles-Rubella (MR) and Inactivated Poliovirus Vaccine (IPV).
- VVM 14 (Medium Heat Stability): Reaches discard point in 14 days at 37°C. Used for Bacillus Calmette-Guerin (BCG).
- VVM 30 (High Heat Stability): Reaches discard point in 30 days at 37°C. Used for Pentavalent, Hepatitis B, DPT, Tetanus and adult Diphtheria (Td), and Pneumococcal Conjugate Vaccine (PCV).
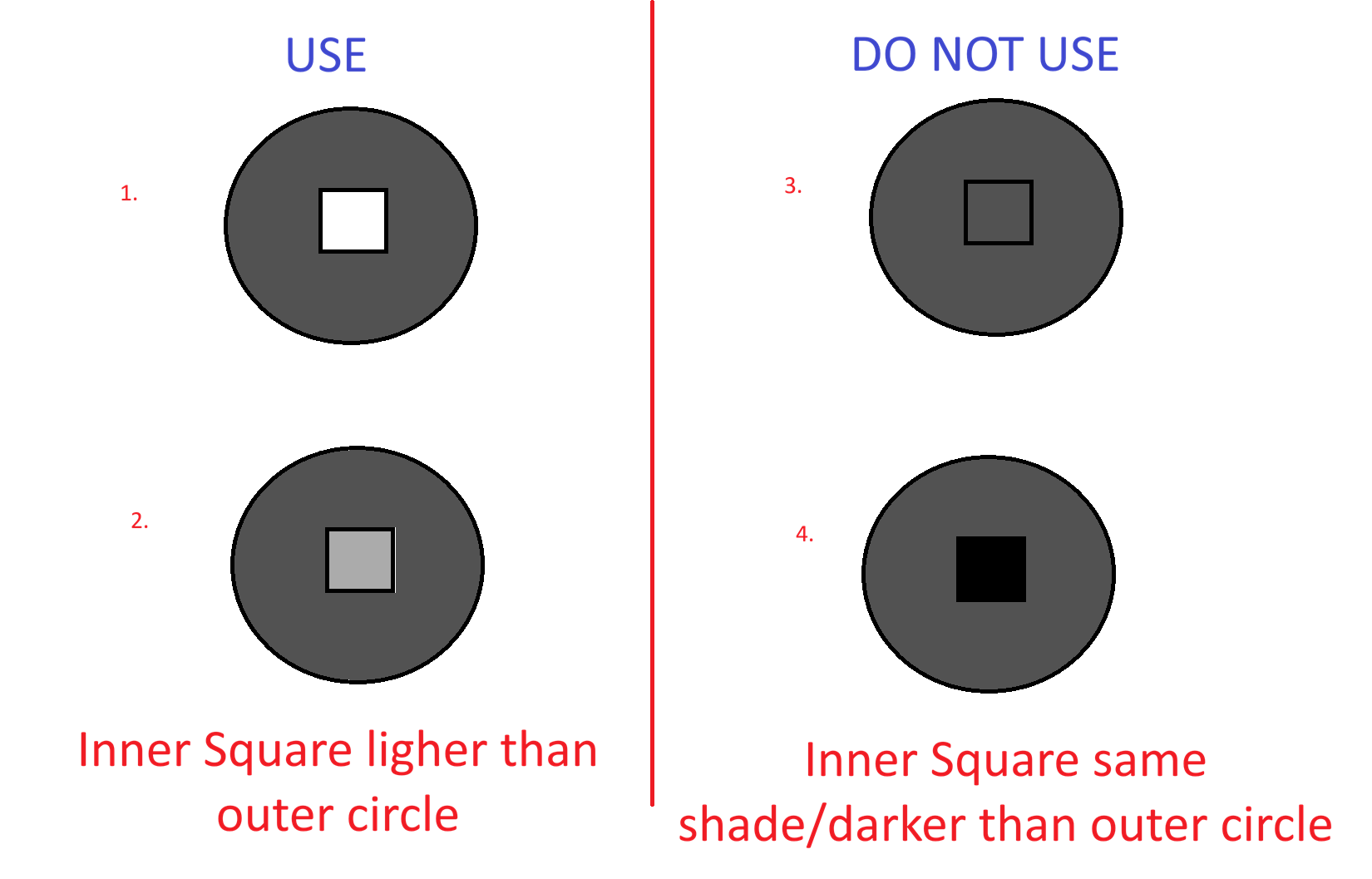
Visual Interpretation Stages
Interpretation relies on comparing the active inner square to the static outer reference ring.
- Stage 1 (Usable): Inner square is completely white or significantly lighter than the outer ring.
- Stage 2 (Usable): Inner square begins to darken but remains distinctly lighter than the outer ring.
- Stage 3 (Discard Point): Inner square is exactly the same color as the outer ring. The vial must be discarded immediately.
- Stage 4 (Beyond Discard): Inner square is darker than the outer ring. The vial must be discarded immediately.
Clinical and Programmatic Significance
- Cold Chain Monitoring: Acts as the only continuous, vial-specific monitor of cumulative heat exposure during transport, storage, and session use.
- Wastage Reduction: Allows healthcare workers to safely use vaccines even if minor, undocumented cold chain excursions have occurred, provided the VVM is in Stage 1 or 2, thereby minimizing unnecessary discard.
- Open Vial Policy (OVP) Integration: VVM status is a mandatory prerequisite for utilizing multi-dose vials for up to 28 days post-opening. A VVM reaching Stage 3 overrides the 28-day allowance, mandating immediate discard.
Limitations
- VVMs measure only heat exposure; they do not indicate exposure to freezing temperatures (freeze-sensitive vaccines still require the Shake Test).
- They do not indicate physical damage, contamination, or whether the manufacturer’s expiration date has passed.
